Full HTML
A Clinical Review of Continuous Renal Replacement Therapy: Modalities, Indications, and Management in Critical Care
Elmukhtar Habas1, Ala Habas2, Amnna Rayani3, Khaled Alarabi4
Author Affiliation
1 Professor, Senior Consultant, Hamad Medical Corporation, Hamad General Hospital, Doha, Qatar
2 Medicine Resident, TCH, University of Tripoli, Tripoli, Libya
3 Professor, Senior Consultant, University of Tripoli, Tripoli, Libya
4 Associate Consultant, Hamad Medical Corporation, Hamad General Hospital, Doha, Qatar
Abstract
Continuous renal replacement therapy (CRRT) is a cornerstone of contemporary intensive care for patients experiencing severe acute kidney injury (AKI), especially in cases involving hemodynamic instability or multiorgan dysfunction. In contrast to intermittent modalities, CRRT facilitates gradual and continuous removal of solutes and fluids, thereby enhancing hemodynamic stability and enabling precise metabolic regulation. This review synthesizes existing evidence and practice, outlining the pathophysiology of AKI in critical illness and the technical specifications of core CRRT modalities: Continuous Venovenous Hemofiltration (CVVH), Continuous Venovenous Hemodialysis (CVVHD), and Continuous Venovenous Hemodiafiltration (CVVHDF). It analyzes the evidence-based indications and optimal timing for initiation, highlighting the shift from early empirical use to a strategy guided by specific clinical criteria. The review delineates fundamental operational principles, encompassing dosing (20–25 mL/kg/h effluent), fluid management, and the pivotal function of regional citrate anticoagulation. The prevention and management of complications, particularly electrolyte disturbances such as hypophosphatemia, nutrient depletion, and altered drug pharmacokinetics, is a primary focus. The analysis focuses on key controversies surrounding the application of CRRT for blood purification in sepsis. This discussion situates CRRT within the wider framework of organ support, emphasizing future advancements in personalized medicine, integrated sorbent technologies, and wearable devices. This review outlines a framework for clinicians to enhance CRRT delivery and improve outcomes for critically ill patients, while recognizing the limitations in the evidence base and global accessibility.
DOI: 10.63475/yjm.v4i3.0259
Keywords: Continuous renal replacement therapy (CRRT), acute kidney injury (AKI), critical care, hemodiafiltration, regional citrate anticoagulation, drug dosing, metabolic complications
Pages: 492-506
View: 24
Download: 31
DOI URL: https://doi.org/10.63475/yjm.v4i3.0259
Publish Date: 31-12-2025
Full Text
Continuous renal replacement therapy (CRRT) represents a cornerstone of modern intensive care for patients with severe acute kidney injury (AKI). Unlike intermittent modalities, CRRT provides gradual, continuous, and physiologically gentle clearance of solutes and fluids. Its capacity for precise volume control while maintaining hemodynamic stability makes it the preferred modality for critically ill patients with hemodynamic instability, multiorgan dysfunction, or refractory circulatory shock. [1] The clinical necessity for this therapy is underscored by the immense global burden of AKI, a syndrome affecting approximately 13.3 million individuals annually and directly contributing to an estimated 1.7 million deaths. [2] This burden exhibits stark regional disparities shaped by variations in healthcare infrastructure and prevalent etiologies.
In Europe, AKI incidence ranges from 10% to 20% in general hospital admissions to over 50% in intensive care unit (ICU) populations, with associated mortality rates of 20% to 50%. [3,4] In the Middle East and North Africa (MENA) region, AKI is often community-acquired, linked to volume depletion, sepsis, and nephrotoxin exposure, with reported ICU mortality ranging from 35% to 60%. [3,4] Research from Asia reveals a comparable burden, with studies from China and India showing AKI occurs in 20% to 30% of ICU patients and is a significant independent predictor of mortality. [3,4] In Africa, epidemiology is characterized by infectious diseases (e.g., malaria, diarrheal illnesses), obstetric complications, and untreated chronic kidney disease, with hospital mortality rates often exceeding 50% due to late presentation and restricted access to renal replacement therapy (RRT).
Within this context of a high global burden and stark inequities in access, CRRT has emerged as a crucial tool for managing severe AKI. However, its utilization mirrors global health inequities. In well-resourced healthcare systems, CRRT may constitute over 70% of all RRT initiated in the ICU for hemodynamically unstable patients. [5] were on chronic dialysis, or had kidney transplant or if their length of hospital stay was <24 hrs. Interventions: None. Measurements and main results: We included 20,126 patients. Mean age was 64 yrs, 14.7% of patients required intensive care unit admission, and hospital mortality was 8.0%. According to the RIFLE criteria, 9.1% of all patients were in the Risk category for acute renal failure, 5.2% were in the Injury category, and 3.7% were in the Failure category. There was an almost linear increase in hospital mortality from Normal to Failure (Normal, 4.4%; Risk, 15.1%; Injury, 29.2%; and Failure, 41.1% Its application remains constrained across much of Asia, Africa, and the MENA region by substantial barriers, including cost, technical complexity, and a scarcity of trained personnel. [2–4]
This review synthesizes evidence identified through a targeted literature search conducted in December 2025. The highest priority was assigned to evidence from large, multicenter randomized controlled trials (RCTs) and individual patient data meta-analyses (e.g., STARRT-AKI, RENAL, and ATN trials). Where high-level RCT evidence was absent or inconclusive, findings from well-designed prospective observational cohorts, registry studies, and international consensus guidelines from expert societies (e.g., Kidney Disease: Improving Global Outcomes [KDIGO], Acute Disease Quality Initiative [ADQI], and European Society for Clinical Nutrition and Metabolism [ESPEN]) were incorporated. For technical and mechanistic details, pivotal physiological studies and manufacturer data were also considered.
To capture developments following pivotal trials (e.g., 2018–2025), the search focused primarily on publications from January 1, 2020, to December 30, 2025. Searches were performed in the biomedical databases PubMed/MEDLINE, Scopus, and the Cochrane Central Register of Controlled Trials (CENTRAL). A structured search strategy employed a combination of Medical Subject Headings (MeSH) and relevant keywords, including: “Renal Replacement Therapy” [MeSH] OR “Continuous Renal Replacement Therapy” OR CRRT OR CVVH OR CVVHD OR CVVHDF AND (“Acute Kidney Injury” [Mesh] OR “Acute Renal Failure”) AND (modalities OR indication OR prescription OR anticoagulation OR citrate OR heparin OR dosing OR complication OR outcome).
A comprehensive understanding of the pathophysiology of AKI is essential for grasping the rationale behind CRRT and its implementation. AKI in the ICU is seldom an isolated occurrence; rather, it is generally part of multiple organ dysfunction syndrome (MODS), influenced by a complex interaction of hemodynamic, inflammatory, and toxic factors. [5] were on chronic dialysis, or had kidney transplant or if their length of hospital stay was <24 hrs. Interventions: None. Measurements and main results: We included 20,126 patients. Mean age was 64 yrs, 14.7% of patients required intensive care unit admission, and hospital mortality was 8.0%. According to the RIFLE criteria, 9.1% of all patients were in the Risk category for acute renal failure, 5.2% were in the Injury category, and 3.7% were in the Failure category. There was an almost linear increase in hospital mortality from Normal to Failure (Normal, 4.4%; Risk, 15.1%; Injury, 29.2%; and Failure, 41.1%.
Mechanisms of AKI
The traditional classification of prerenal, intrinsic, and postrenal AKI remains useful, but in critical illness, these categories frequently overlap. [1]
• Hemodynamic Mediated (Prerenal and Ischemic): Systemic hypotension, decreased cardiac output, or intrarenal vasoconstriction (due to vasopressors, sepsis, or hepatorenal syndrome) can all result in a significant reduction in renal blood flow and glomerular filtration rate (GFR). [6] This triggers a sequence of cellular hypoxia, ATP depletion, and inflammation. The outer medulla is characterized by high metabolic demand and a precarious blood supply, making it a primary site of injury.
• Inflammatory and Septic AKI: Sepsis constitutes the primary etiology of AKI in the ICU. The pathophysiology extends beyond hypoperfusion to include a dysregulated host response. Essential mechanisms comprise:
― Macrocirculatory and Microcirculatory Dysfunction: Despite restored systemic pressure, microvascular shunting and endothelial dysfunction can persist, leading to heterogeneous perfusion and continued tubular ischemia. [6]
― Immune and Inflammatory Activation: The release of damage-associated molecular patterns (DAMPs) and pathogen-associated molecular patterns (PAMPs) initiates a cytokine storm. The inflammatory environment leads to direct injury of tubular epithelial cells, apoptosis, and infiltration of leukocytes. [6]
― Metabolic Reprogramming: Tubular cells may alter their metabolic processes to endure hypoxia; however, this adaptation can increase their vulnerability to additional injury.
• Nephrotoxic: Patients in the ICU encounter various nephrotoxins, such as aminoglycosides, vancomycin, iodinated contrast media, and myoglobin associated direct tubular cytotoxicity, lead to intra-tubular obstruction, or further aggravate ischemic injury.
The Clinical Spectrum and Staging
In all cases, the severity of AKI is categorized according to the KDIGO criteria, which take into account variations in serum creatinine and urine output [8,9]:
• Stage 1: Serum creatinine increase ≥0.3 mg/dL (≥26.5 μmol/L) or to 1.5 to 1.9 times baseline, or urine output <0.5 mL/kg/h for 6 to 12 hours.
• Stage 2: Creatinine 2.0 to 2.9 times baseline, or urine output <0.5 mL/kg/h for ≥12 hours.
• Stage 3: Creatinine increase to ≥4.0 mg/dL (≥353.6 μmol/L), initiation of RRT, or urine output <0.3 mL/kg/h for ≥24 hours or anuria for ≥12 hours.
The staging exhibits prognostic significance, as higher stages are associated with elevated mortality rates and extended periods of organ support. [5] were on chronic dialysis, or had kidney transplant or if their length of hospital stay was <24 hrs. Interventions: None. Measurements and main results: We included 20,126 patients. Mean age was 64 yrs, 14.7% of patients required intensive care unit admission, and hospital mortality was 8.0%. According to the RIFLE criteria, 9.1% of all patients were in the Risk category for acute renal failure, 5.2% were in the Injury category, and 3.7% were in the Failure category. There was an almost linear increase in hospital mortality from Normal to Failure (Normal, 4.4%; Risk, 15.1%; Injury, 29.2%; and Failure, 41.1% Serum creatinine is a delayed and imperfect biomarker in this setting, as it is influenced by volume status, muscle mass, and tubular secretion. This has prompted investigations into functional biomarkers (e.g., furosemide stress test) and damage biomarkers (e.g., [TIMP-2] · [IGFBP7]) to enhance early diagnosis and risk stratification. [10,11] While currently primarily prognostic, the ongoing evolution of these biomarkers holds promise for more objectively informing critical decisions, such as the optimal timing for RRT initiation, a key controversy discussed later in this review.
From Injury to Support: The Rationale for CRRT
The ramifications of severe AKI extend beyond the impairment of excretory function. The impaired kidney induces a proinflammatory condition, exacerbates fluid overload (resulting in tissue edema, compromised organ perfusion, and heightened intra-abdominal pressure), and disturbs immune homeostasis. [5,12] were on chronic dialysis, or had kidney transplant or if their length of hospital stay was <24 hrs. Interventions: None. Measurements and main results: We included 20,126 patients. Mean age was 64 yrs, 14.7% of patients required intensive care unit admission, and hospital mortality was 8.0%. According to the RIFLE criteria, 9.1% of all patients were in the Risk category for acute renal failure, 5.2% were in the Injury category, and 3.7% were in the Failure category. There was an almost linear increase in hospital mortality from Normal to Failure (Normal, 4.4%; Risk, 15.1%; Injury, 29.2%; and Failure, 41.1% The objectives of CRRT extend beyond replacing glomerular filtration to include: (1) Offering multiorgan support through the correction of lifethreatening metabolic and fluid imbalances associated with AKI. (2) Reducing the systemic inflammatory burden via the convective clearance of middle-molecular-weight mediators (the “peak concentration hypothesis”). [13] (C) Creating a stable physiological environment to promote the recovery of native renal function and other organ systems. This pathophysiological basis underpins all subsequent decisions related to CRRT modality, timing, and intensity.
The acronym CRRT refers to a collection of several modalities, each of which is distinguished by the fundamental mechanism that it employs for solute clearance and fluid control. The selection among these options determines the effectiveness of removing solutes of varying molecular weights and affects operational parameters. All modalities necessitate vascular access through a double-lumen central venous catheter and are operated by a peristaltic blood pump. [14]
Continuous Venovenous Hemofiltration (CVVH)
• Primary Mechanism: Convection. Solute removal occurs through the bulk movement of plasma water across a high-flux, porous membrane, driven by a transmembrane pressure (TMP) gradient. Ultrafiltration involves the movement of dissolved solutes alongside water, a phenomenon known as “solvent drag.” [15,16]
• The ultrafiltrate generated is primarily plasma water, encompassing all solutes capable of traversing the membrane pores.
• Operational Setup:
― Replacement Fluid: To avert critical volume depletion, the ultrafiltrate volume is substituted with a sterile, balanced electrolyte solution. Infusion can occur either pre-filter (pre-dilution) or post-filter (post-dilution). [17]
― Pre-dilution vs. Post-dilution: In pre-dilution, replacement fluid is introduced before the filter, diluting the blood. This minimizes hemoconcentration in the filter, thereby decreasing the risk of clot formation and potentially extending the filter’s lifespan; however, it also leads to a dilution of solute concentration, which diminishes clearance efficiency per liter of exchange. [17] Postdilution infusion takes place subsequent to the filter, enhancing clearance efficiency while elevating the risk of filter clotting as a result of hemoconcentration at the distal end of the filter. • Clearance Profile: Demonstrates high efficacy for middle-molecular-weight substances ranging from 500 to 20,000 Da, encompassing inflammatory cytokines such as IL-6 and TNF-α, myoglobin, and certain antibiotics. [7,17] Small solute clearance, such as urea and potassium, exhibits lower efficiency per liter of effluent when compared to diffusive techniques. • Prescription: The dose represents the effluent rate, defined as the sum of the ultrafiltration rate and the replacement fluid rate. The standard dosage ranges from 25 to 35 mL/kg/h of effluent. [18]
Continuous Venovenous Hemodialysis (CVVHD)
• Primary Mechanism: Diffusion is the primary mechanism in this process. Solute removal transpires along a concentration gradient through a semipermeable membrane. Dialysate is a sterile solution that maintains electrolyte concentrations similar to physiological plasma levels and flows counter-current to blood on the opposite side of the membrane. [15,16] Small solutes undergo diffusion from regions of higher concentration (blood) to regions of lower concentration (dialysate).
• Operational Setup:
― Dialysate Flow: Small solute clearance is primarily determined by the rate of dialysate flow, which is typically between 15 and 40 mL/ kg/h and is controlled by a pump rotation rate.
― Ultrafiltration: Fluid removal is regulated independently by a pump that creates a TMP, resulting in the formation of an ultrafiltrate. No replacement fluid is utilized for solute clearance.
• Clearance Profile: Highly effective for small solutes (less than 500 Da), including urea, creatinine, potassium, and various ions. The removal of larger molecules is relatively inefficient. [15]
• Prescription: The dosage is established based on the dialysate flow rate and the ultrafiltration rate to maintain fluid balance. [18]
Continuous Venovenous Hemodiafiltration (CVVHDF)
• Primary Mechanism: Combined Convection and Diffusion. CVVHDF effectively integrates the methodologies of CVVH and CVVHD, utilizing dialysate for diffusion alongside concurrent replacement fluid for convection. [15]
• Operational Setup: This setup is characterized by its complexity, necessitating the use of both dialysate and replacement fluid pumps. Either pre-dilution or postdilution can be applied alongside the dialysate flow.
• Clearance Profile: Delivers the widest and most effective solute clearance throughout the molecular weight range. The method provides excellent small solute clearance through diffusion and improved removal of middle molecules via convection. [15,16] Therefore, it is often considered the default modality in modern ICUs with sufficient resources, offering comprehensive solute control. In practice, however, the optimal modality must balance theoretical efficacy with local logistics, cost, and protocol familiarity.
• Prescription: The overall effluent dose comprises the combined flows of ultrafiltrate, dialysate, and replacement fluid. [18]
Slow Continuous Ultrafiltration (SCUF)
• Primary Mechanism: SCUF is exclusively utilized for fluid removal. A notably slow ultrafiltration rate (generally ranging from 100 to 500 mL/h) is utilized without incorporating replacement fluid or dialysate. [19] As a result, the clearance of solute is minimal.
• Indication: Its main function is to address refractory fluid overload in patients who primarily maintain renal solute clearance, as seen in conditions like decompensated heart failure or hepatorenal syndrome. [19] Table 1 summarizes key aspects of these CRRT modalities.
Table 1: Comparison of core continuous renal replacement therapy (CRRT) modalities.
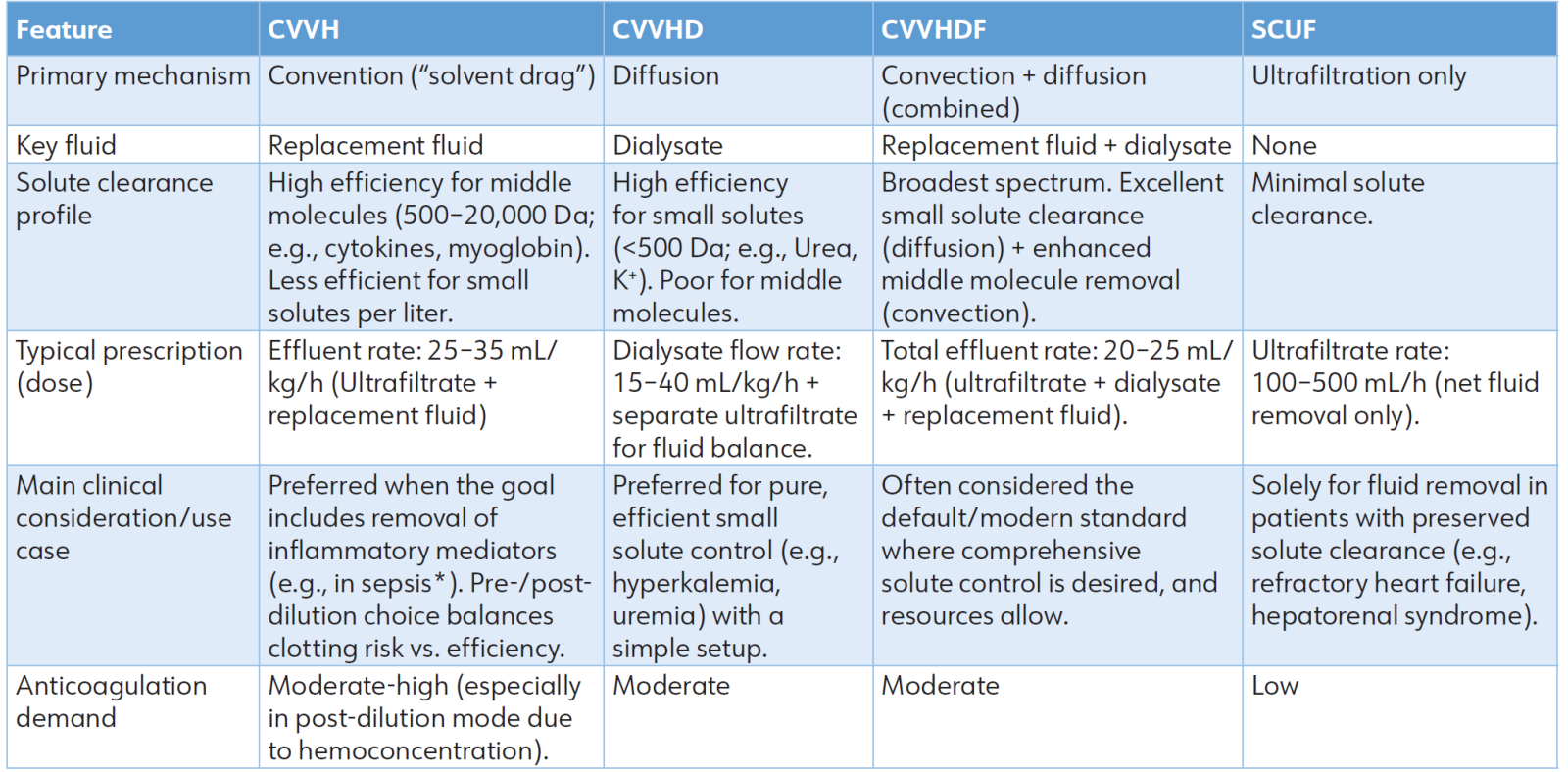
The use of CRRT for immunomodulation in sepsis remains controversial and is not supported by mortality benefit in unselected populations (see Sections 4.2 and 10.1).
CVVH: Continuous Venovenous Hemofiltration; CVVHD: Continuous Venovenous Hemodialysis; CVVHDF: Continuous Venovenous Hemodiafiltration; SCUF: Slow Continuous Ultrafiltration.
The choice to commence CRRT stands as a pivotal and intricate aspect within the realm of critical care nephrology. Although absolute indications are well-defined, many decisions take place in a nuanced “grey zone,” necessitating a thoughtful combination of clinical, laboratory, and physiological factors. The ideal timing for initiation continues to be a subject of ongoing investigation and discussion.
Absolute (Traditional) Indications
These life-threatening complications of AKI require prompt RRT, regardless of whether CRRT or intermittent hemodialysis (IHD) is selected. [8,20]
• Refractory Hyperkalemia: Serum potassium levels exceeding 6.5 mmol/L or accompanied by electrocardiographic changes (such as peaked T waves or QRS widening) that do not respond to medical interventions (including insulin-dextrose, salbutamol, or sodium bicarbonate).
• Refractory Metabolic Acidosis: Characterized by severe acidemia (pH <7.15) primarily resulting from AKI, leading to hemodynamic instability or compromised cardiac contractility, and showing no response to bicarbonate therapy.
• Refractory Fluid Overload: The presence of pulmonary edema or anasarca resulting in respiratory or cardiac compromise, which does not respond to diuretic therapy. The ability of CRRT to facilitate gentle, continuous fluid removal is a notable advantage. [21]
• Uremic Complications: Encephalopathy, pericarditis, bleeding diathesis, or other clinical manifestations directly linked to the buildup of uremic toxins.
• Intoxications with Dialyzable Toxins: This category includes specific toxins characterized by low molecular weight, low volume of distribution, and low protein binding, such as lithium, methanol, ethylene glycol, and salicylate. [15]
Relative Indications and Broader Rationale in Critical Illness
In addition to these traditional indications, CRRT is frequently started for multisystem support in ICU patients. The objective is to avert complications and establish a conducive environment for organ recovery. [22]
• Support for Additional Treatments: To ensure sufficient nutrition, administration of blood products, or delivery of intravenous medications in patients with anuria or oliguria who have limited fluid tolerance.
• Electrolyte and Acid-Base Management: Significant, advancing dysnatremias (hyper- or hyponatremia) necessitating regulated correction. CRRT enables meticulous control of sodium balance by tailoring the composition of dialysate and replacement fluids. • Thermoregulation: Utilized as a supplementary approach in cases of severe, refractory hyperthermia (e.g., malignant hyperthermia, heat stroke).
• Support in Sepsis/MODS (Blood Purification): The concept of using high-volume hemofiltration or other extracorporeal techniques to modulate the inflammatory cascade by removing mid-molecularweight mediators remains a topic of investigation and debate. While some studies and early trials (e.g., the IVOIRE trial) have shown transient improvements in hemodynamic parameters, larger RCTs (e.g., the COMPACT trial) have failed to demonstrate a consistent mortality benefit for unselected populations. [13,23] Therefore, the use of CRRT specifically for immunomodulation in sepsis is currently regarded as experimental and is not recommended outside of defined research protocols.
The Timing Debate: “Early” vs. “Late” Initiation
• Timing of initiating RRT: Without a definitive indication, the timing of initiating RRT has been explored in various significant trials, which present seemingly contradictory findings, contributing to a more refined modern perspective. [18,24,25]
• Key Trials: In the ELAIN study conducted in 2016, a single-center trial involving a cardiac surgery cohort indicated a potential mortality benefit associated with the “early” initiation of treatment at KDIGO stage 2, as opposed to “late” initiation at KDIGO stage 3. [26] Furthermore, the multicenter AKIKI and STARRT-AKI trials observed that an accelerated RRT strategy did not yield any mortality benefits. Additionally, potential harms were noted, such as a rise in catheter-related complications, with no significant difference in renal recovery outcomes. [24,25]
• Current Consensus and Pragmatic Approach: A definitive individual patient data meta-analysis synthesizing the results of these major trials [24] supports a “watchful waiting” or “standard” strategy for the majority of patients. [18] This approach entails proactive medical management of AKI with vigilant monitoring for the emergence of absolute indications or a clear clinical trajectory necessitating RRT. Initiation is justified when one or more definitive criteria are met, or when a combination of significant and worsening biochemical derangements, progressive fluid overload, and clinical decline strongly suggests an imminent requirement.
• Biomarker-Guided Strategies: The future of timing could be rooted in personalized medicine. Biomarker panels (e.g., NephroCheck®) could assist in identifying patients with subclinical tubular damage who are at elevated risk for progression, potentially enabling more targeted intervention. [10,11]
Figure 1 illustrates a pragmatic clinical decision pathway for CRRT initiation, synthesizing evidence from major trials (STARRT-AKI, AKIKI) and KDIGO recommendations.
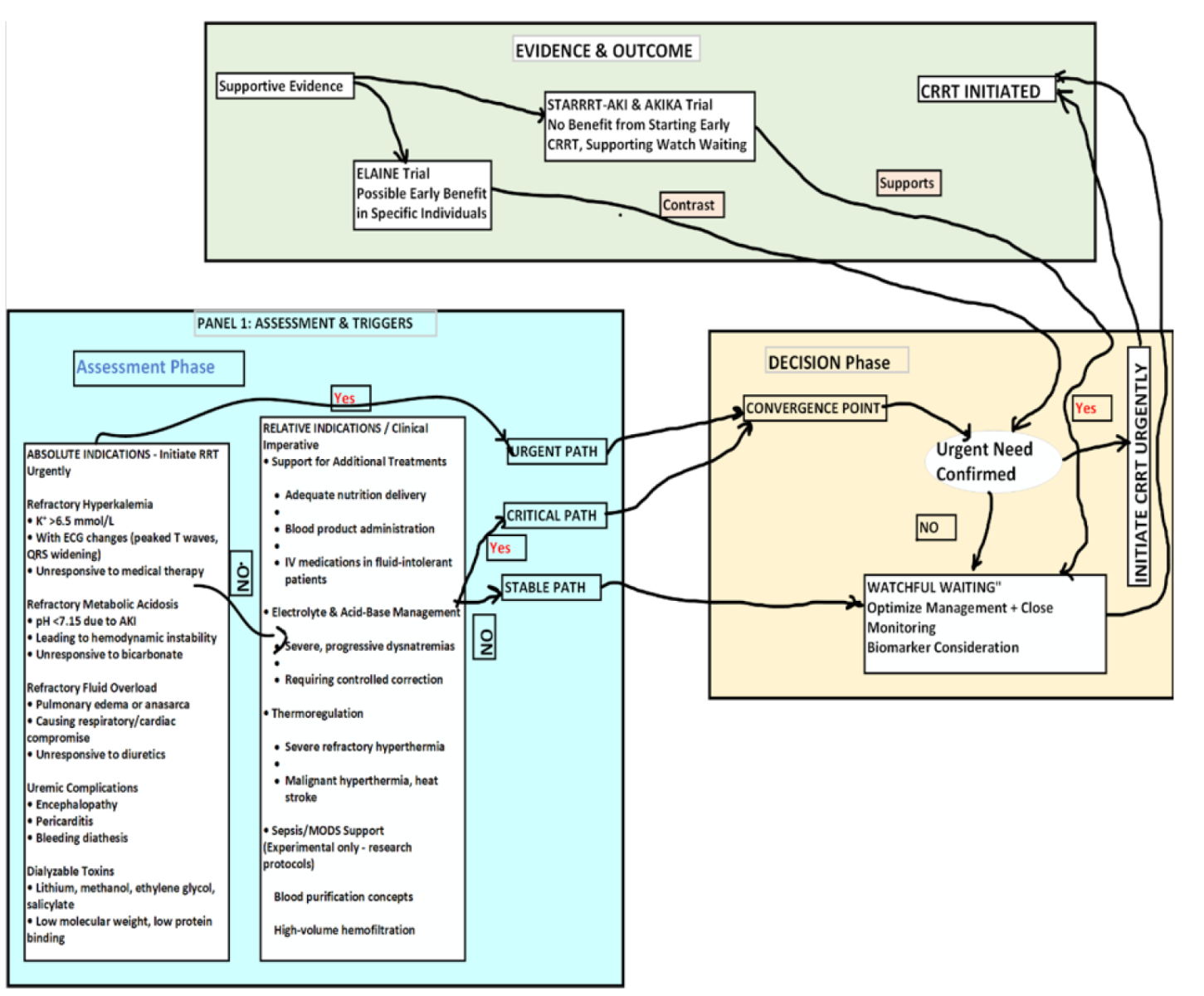
Figure 1: Algorithm for the assessment and decision to initiate continuous renal replacement therapy (CRRT) in acute kidney injury, integrating evidence from major clinical trials and absolute/relative clinical indications.
The effective implementation of CRRT relies on a consistent methodology for circuit management, precise dose prescription, and real-time modifications in response to patient feedback. Contemporary CRRT devices incorporate advanced safety mechanisms and fluid balance management, yet the importance of clinical expertise cannot be overstated.
Core Components of the CRRT Circuit
A standard circuit consists of several integrated subsystems [15,27]:
A. Vascular Access: A specialized, large-bore (11.5– 14 Fr) double-lumen catheter is crucial. The right internal jugular vein is the preferred site for achieving superior flow rates and minimizing the risk of infection, with the femoral and subclavian veins being secondary options. Catheter dysfunction, including issues such as kinking, malposition, and fibrin sheath formation, significantly contributes to therapy downtime.
B. Extracorporeal Circuit Tubing: Establishes the connection between the patient and the filter. Contemporary circuits frequently undergo surface treatment to improve biocompatibility and minimize thrombogenicity.
C. Blood Pump: Produces the extracorporeal blood flow rate (QB). The typical range for QB is between 100 and 200 mL/min. Increased flows exceeding 150 mL/min enhance the efficiency and durability of thoe filter; hwever, they may not be well-tolerated in cases of severe shock.
D. Hemofilter/Dialyzer: The semipermeable membrane facilitates the exchange of solutes and fluids. Contemporary high-flux filters utilize synthetic membranes, such as polysulfone and polyamide, featuring extensive surface areas ranging from 1.2 to 1.8 m² and specific pore sizes to facilitate effective convective and diffusive transport. [16]
E. Fluid Management System: The core of CRRT accuracy. The components include: ― Anticoagulant Pump: Delivers citrate or heparin before filtration. ― Replacement Fluid Pump(s): Intended for CVVH/ CVVHDF applications. ― Dialysate Pump: Designed for Continuous Venovenous Hemodialysis and Continuous Venovenous Hemodiafiltration. ― Effluent Pump: Extracts the complete ultrafiltrate (waste fluid). The computer system of the machine consistently regulates all inputs and outputs to maintain the specified net fluid balance.
F. Safety Monitors: Incorporate air detectors, pressure sensors (pre-filter, post-filter, transmembrane), and blood leak detectors that will stop the pump if a critical threshold is exceeded.
Prescription and Dose Calculation
The dose of CRRT is measured by the total effluent rate, which indicates the volume of plasma water cleared over a specified time period. This is adjusted according to the patient’s body weight. [28,29]
• Effluent Rate (Qeff): In the context of CVVH and CVVHDF, Qeff is calculated as the sum of the Ultrafiltration Rate and the Replacement Fluid Rate, with the addition of the Dialysate Rate for CVVHDF. In CVVHD, the clearance of small solutes is mainly influenced by the flow rate of the dialysate (Qd).
• Prescribed Dose: According to significant clinical studies (ATN, RENAL), the typical recommended dosage is 20 to 25 mL/kg/h of effluent. [28,29] To calculate this dose, an ideal or adjusted body weight should be used for patients with obesity, rather than actual body weight, to avoid significant under-dosing. Administering a higher dose (e.g., 35– 40 mL/kg/h) has not shown enhanced survival rates and may elevate the risks of nutrient depletion and electrolyte imbalances. [28]
• Delivered Dose: It is essential to differentiate between the prescribed dose and the delivered dose. Therapy often faces interruptions due to procedures, circuit clotting, or machine alarms. The delivered dose frequently falls short of the prescribed amount by 10% to 30%. Tracking the actual mean effluent rate over 24 hours serves as an essential quality metric. [30]
Key Operational Parameters and Adjustments
Anticoagulation Protocol: As outlined in Section 7, the selection (RCA vs. heparin) has a direct impact on filter longevity and the risk of bleeding. [31,32]
• Fluid Balance Management: This represents an evolving, therapeutic objective. A standard methodology includes:
― Assessment: Determine the overall daily fluid balance by subtracting the total outputs from the total inputs.
― Goal: To establish a net negative fluid balance for patients experiencing fluid overload (for instance, −100 to −300 mL/h), maintain a net even balance for those who are euvolemic, or implement a net positive balance in specific instances of hypovolemia.
― Adjustment: Frequently reassess fluid status (e.g., every 4–6 hours) and modify the net balance prescription as needed. [17]
• Customizing Fluid Composition: Conventional commercial fluids possess predetermined electrolyte compositions. In cases of intricate metabolic disturbances (such as severe hypophosphatemia and hypernatremia), the electrolyte composition of replacement fluid or dialysate can be tailored in collaboration with pharmacy, which is fundamental to precision CRRT. [18]
Monitoring and Troubleshooting
Successful CRRT necessitates careful observation of the patient and the circuit.
• Patient Monitoring: Regular evaluation of hemodynamics, electrolytes (notably potassium, phosphate, magnesium, and ionized calcium with RCA), acid-base balance, and clinical signs of fluid overload or depletion.
• Circuit Monitoring: ― Pressures: Increased TMP or pre-filter pressure frequently suggests the occurrence of filter clotting. A decrease in venous pressure could indicate inadequate blood return or potential complications with the catheter.
― Filter Clotting: It represents the predominant reason for the failure of therapy. To address this issue, it is important to implement strategies such as ensuring sufficient anticoagulation, striving to maintain a QB greater than 150 mL/min when feasible, avoiding excessive hemoconcentration (with post-filter Hct exceeding 65%), and reducing unnecessary circuit interruptions. [28]
― Citrate Toxicity: During RCA, it is essential to monitor the serum total calcium to ionized calcium ratio to assess citrate toxicity. A ratio exceeding 2.5 indicates citrate accumulation and necessitates a revision of the protocol. [33] Figure 2 demonstrates a systematic approach to diagnosing and managing common circuit pressure abnormalities.
Figure 2 represents a flow chart for troubleshooting the CRRT circuit alarm and possible solutions.
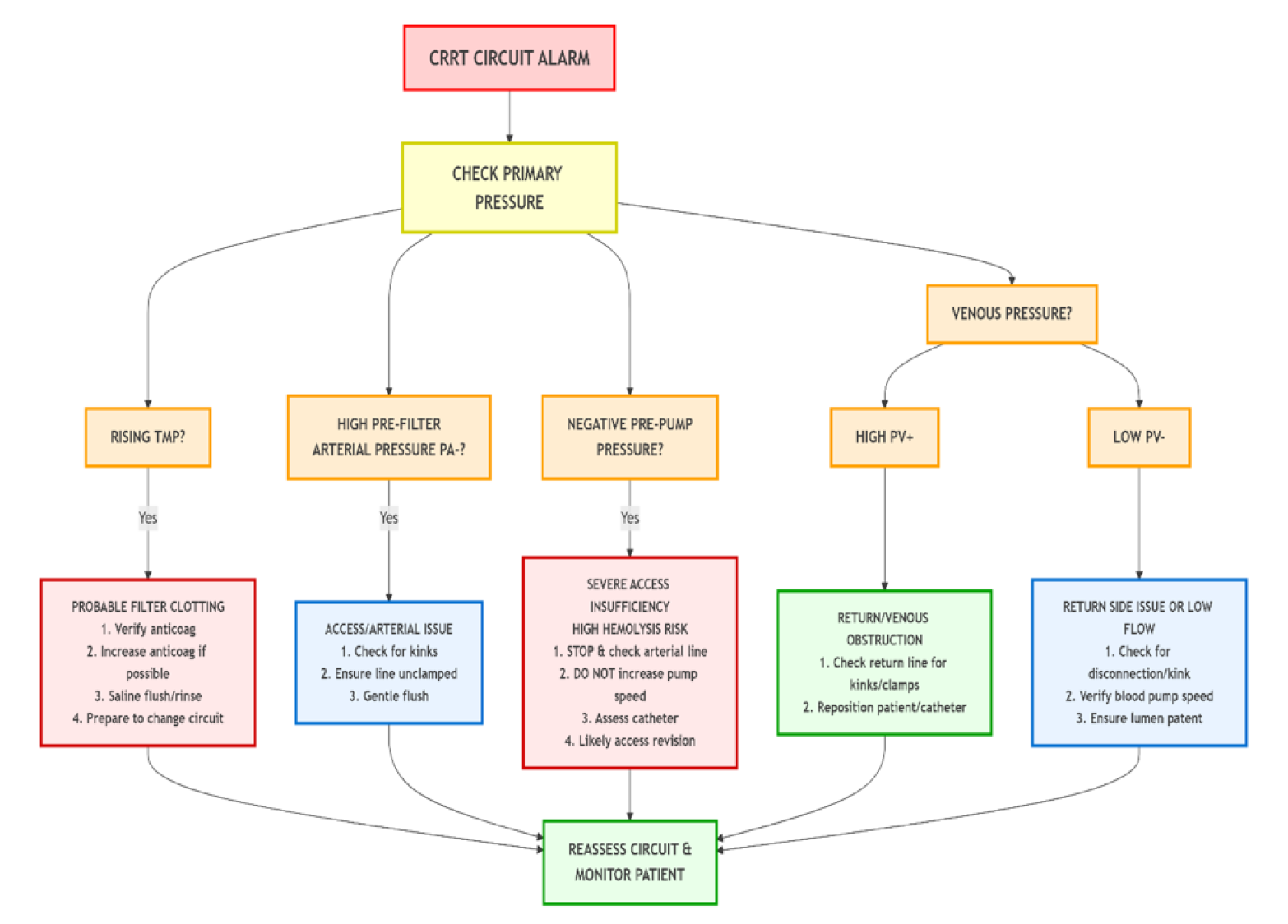
Figure 2: Clinical decision pathway for troubleshooting CRRT circuit alarms. TMP: transmembrane pressure; PV: venous pressure; PA: arterial pressure.
Ensuring effective anticoagulation of the extracorporeal circuit is crucial for maintaining filter patency, facilitating adequate therapy delivery, and reducing blood loss. The selection of an anticoagulant approach is a vital factor influencing the effectiveness and safety of CRRT, necessitating a meticulous consideration of circuit durability alongside the individual bleeding risk of the patient. [31,34]
Primary Anticoagulation Strategies
Regional Citrate Anticoagulation (RCA) RCA has emerged as the primary and favored anticoagulation approach for the majority of CRRT patients who do not have significant contraindications, bolstered by meta-analyses that highlight its advantages in circuit longevity and minimizing bleeding risk. [31,34]
• Mechanism: Trisodium citrate is introduced before filtration, effectively chelating ionized calcium (Ca²+) within the extracorporeal circuit, which in turn inhibits the calcium-dependent coagulation cascade. Systemic anticoagulation is not utilized since citrate is quickly metabolized by the liver, skeletal muscle, and kidney cortex into bicarbonate. Meanwhile, calcium is re-infused through a separate central or post-filter infusion to ensure normal systemic ionized calcium levels. [33]
• Protocols: Contemporary protocols generally aim for a post-filter ionized calcium concentration of 0.25 to 0.35 mmol/L. It is essential to conduct strict monitoring of systemic ionized calcium, sodium, and acid-base status (including anion gap and total Ca²+/ ionized Ca²+ ratio) every 4 to 6 hours at the outset.
• Advantages: Extended circuit longevity (typically 60–70+ hours); markedly lower risk of severe bleeding in comparison to systemic heparin; possible anti-inflammatory properties of citrate.
• Complications and Management:
― Citrate Toxicity (Citrate Lock): Citrate toxicity, also known as citrate lock, arises when the metabolism of citrate is exceeded, which can happen in conditions such as liver failure, severe shock, or mitochondrial dysfunction. The outcome is an increase in citrate levels, indicated by an elevated total-to-ionized calcium ratio (>2.5) and the presence of high-anion-gap metabolic acidosis. Management requires a decrease in the citrate infusion rate alongside an increase in calcium supplementation. [33] Implementing RCA requires substantial infrastructure, including dependable access to ionized calcium testing, specialized calcium and citrate solutions, and clinical expertise to manage its metabolic complications effectively. These requirements pose significant barriers to implementation in resource-limited settings.
― Metabolic Alkalosis: Resulting from the systemic conversion of citrate to bicarbonate.
― Hypernatremia: Occurring due to sodium load associated with trisodium citrate; it may necessitate the administration of lower-sodium replacement fluids.
Systemic Heparin Anticoagulation
• Mechanism: Unfractionated heparin enhances the activity of antithrombin III, leading to the inhibition of thrombin and factor Xa, which results in systemic anticoagulation.
• Protocol: Generally consists of an initial bolus (e.g., 2000–5000 IU) succeeded by a continuous infusion (e.g., 5–10 IU/kg/h), adjusted to achieve a target activated partial thromboplastin time (aPTT) of 1.5 to 2 times the normal baseline.
• Advantages: Low cost, ease of monitoring, and reversibility through the use of protamine sulfate.
• Disadvantages: Elevated risk of hemorrhage; potential for heparin-induced thrombocytopenia (HIT); inconsistent efficacy in patients with antithrombin deficiency; reduced average circuit lifespan relative to RCA.
No Anticoagulation (Heparin-Free)
• Indications: Patients exhibiting active bleeding, significant coagulopathy (INR >2, platelet count <50 × 10⁹/L), recent major surgical procedures, or HIT.
• Strategy: Utilizes elevated blood flow rates (>200 mL/min if tolerated), pre-dilution replacement fluid mode, and periodic saline flushes (e.g., 200– 300 mL every 1–2 hours) to mitigate clotting risks. This approach reduces bleeding risk but frequently leads to a markedly shorter filter lifespan (12– 24 hours) and an elevated nursing workload. [33]
Alternative and Adjunctive Strategies
• Prostacyclin (Epoprostenol): Is primarily utilized as an adjunct in Europe. It inhibits platelet aggregation through the elevation of cAMP levels. Typically administered in low doses (e.g., 2–5 ng/kg/min) alongside reduced-dose heparin to mitigate bleeding risk, or used independently in patients with HIT. Notable adverse effects encompass systemic hypotension and flushing.
• Direct Thrombin Inhibitors (Argatroban, Bivalirudin): Serve as the primary treatment for patients with confirmed HIT who require CRRT. Dosing is intricate, necessitating careful monitoring through aPTT or ecarin clotting time, and presents a considerable bleeding risk owing to systemic anticoagulation and the absence of a reversal agent. [35]
Selection and Monitoring
Anticoagulation strategies should be tailored to the individual patient. RCA is indicated for patients who do not exhibit severe liver impairment or profound shock. Systemic heparin or heparinfree regimens are utilized in cases with specific contraindications to citrate or elevated bleeding risk. Regardless of the method employed, it is essential to monitor circuit pressures, filter appearance, and patient coagulopathy vigilantly.
Although CRRT serves as a vital therapeutic intervention, it carries a notable and unique set of complications. These can be generally classified into two categories: technical, which pertains to circuit-related issues, and patient-related factors. These often arise from the ongoing nature of therapy and the extracorporeal circulation of blood. [36,37]
Technical and Circuit-Related Complications
• Filter Clotting: This is the most common complication, resulting in early termination of therapy, blood loss, and heightened expenses. Factors that contribute to the issue encompass insufficient anticoagulation, reduced blood flow rates, hemoconcentration (notably in post-dilution mode), regular circuit interruptions, and less-than-ideal vascular access. [14,28]
• Circuit Air Embolism: An uncommon yet potentially devastating occurrence. Contemporary devices are integrated with ultrasonic air detection systems that secure the venous line and halt the blood pump upon the identification of air. Careful preparation of the circuit and ensuring all connections are firmly in place are essential steps for prevention. [20]
• Hemolysis: The mechanical destruction of red blood cells may arise from excessive negative pre-pump pressure, which can be caused by catheter stenosis or kinking, a kinked arterial line, or a malfunctioning blood pump. Indicators consist of plasma with a pink hue and a rapid decrease in hematocrit levels. [38]
• Blood Leak: Resulting from a rupture in the hollowfiber membrane, which permits blood to infiltrate the dialysate/effluent compartment. The blood leak detection system of the machine will trigger an alarm and halt the effluent pump. Immediate circuit modification is necessary. [39]
• Fluid Balance Errors: Even with automated systems in place, issues such as programming errors or faults in pressure monitoring can result in unintended rapid fluid removal, potentially leading to hypotension or, conversely, fluid overload. Independent verification of prescription parameters is an essential safety measure. [40] Table 2 presents the troubleshooting guide for common CRRT circuit pressure alarms and abnormalities.
Table 2: Troubleshooting guide for common CRRT circuit pressure alarms and abnormalities.
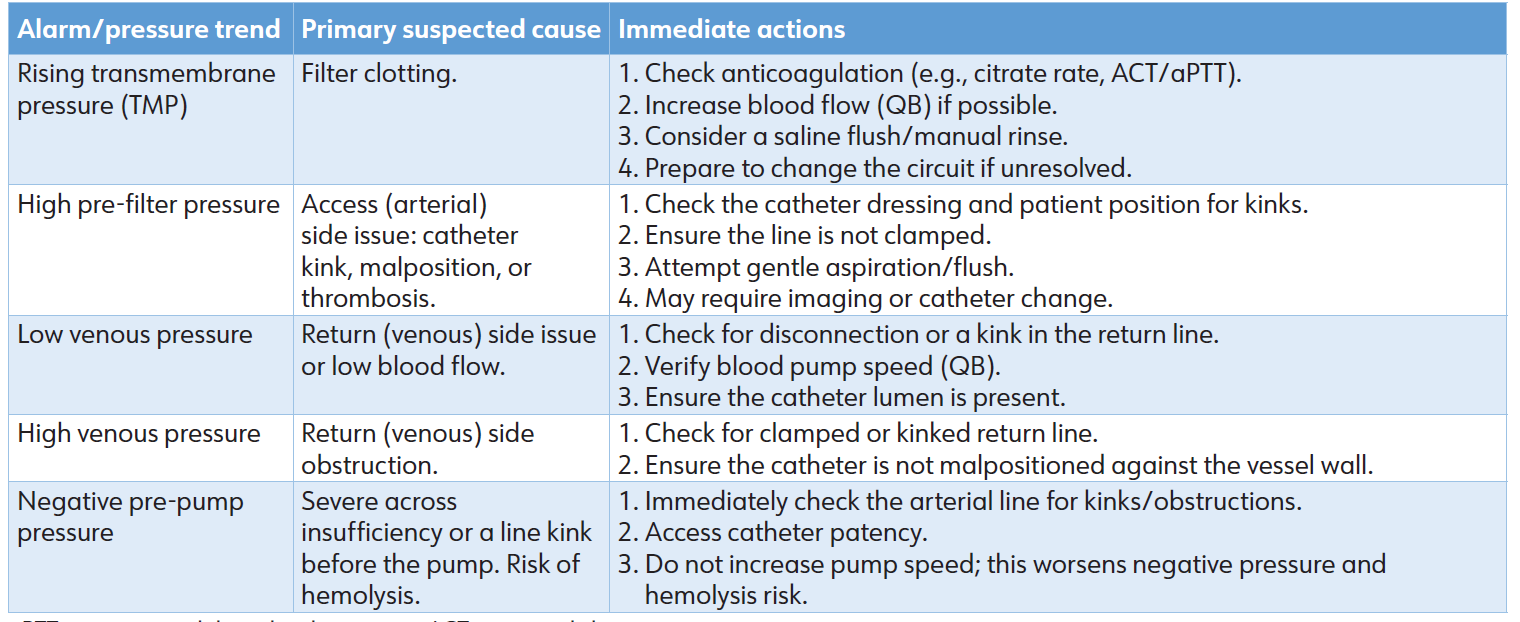
aPTT: active partial thromboplastin time; ACT: activated clotting time.
Patient-Related Metabolic and Electrolyte Complications
The ongoing removal of plasma water results in significant and expected losses.
• Hypophosphatemia: This is one of the most prevalent electrolyte disturbances, occurring in up to 80% of patients undergoing prolonged CRRT. Phosphate undergoes unrestricted filtration, and typical replacement fluids are largely devoid of it. It is essential to implement proactive and protocolized phosphate supplementation, such as intravenous sodium or potassium phosphate, to avert severe depletion. This condition can lead to muscle weakness, respiratory failure, and compromised cardiac function. [41]
• Hypokalemia and Hypomagnesemia: Notable losses are observed, requiring consistent monitoring and supplementation through intravenous or fluidbased replacement.
• Nutrient Depletion: CRRT leads to significant losses of water-soluble vitamins (B1, B6, C, folate) and trace elements (selenium, zinc). Daily intravenous multivitamin and trace element supplementation is a standard practice to prevent deficiency syndromes such as Wernicke›s encephalopathy or refractory lactic acidosis. [42]
• Acid-Base Disorders:
― Metabolic Alkalosis: Frequently observed in RCA due to citrate metabolism to bicarbonate and in high-volume CVVH utilizing lactatebased fluids. Adjustment of fluid composition or citrate dose may be necessary.
― Citrate Toxicity (Metabolic Acidosis): In cases of liver failure or severe shock, the inability to metabolize citrate results in its accumulation, which binds to systemic calcium and induces a high-anion-gap metabolic acidosis. [43]
• Hypothermia: Ongoing exposure of blood to the extracorporeal circuit may result in a decrease in core temperature, particularly at elevated effluent rates. The implementation of integrated blood warmers on the return line is considered standard procedure. Table 3 summarizes common metabolic and electrolyte complications, their mechanisms, and management strategies.
Table 3: Common Metabolic and Electrolyte Complications of CRRT: Mechanism and Management.
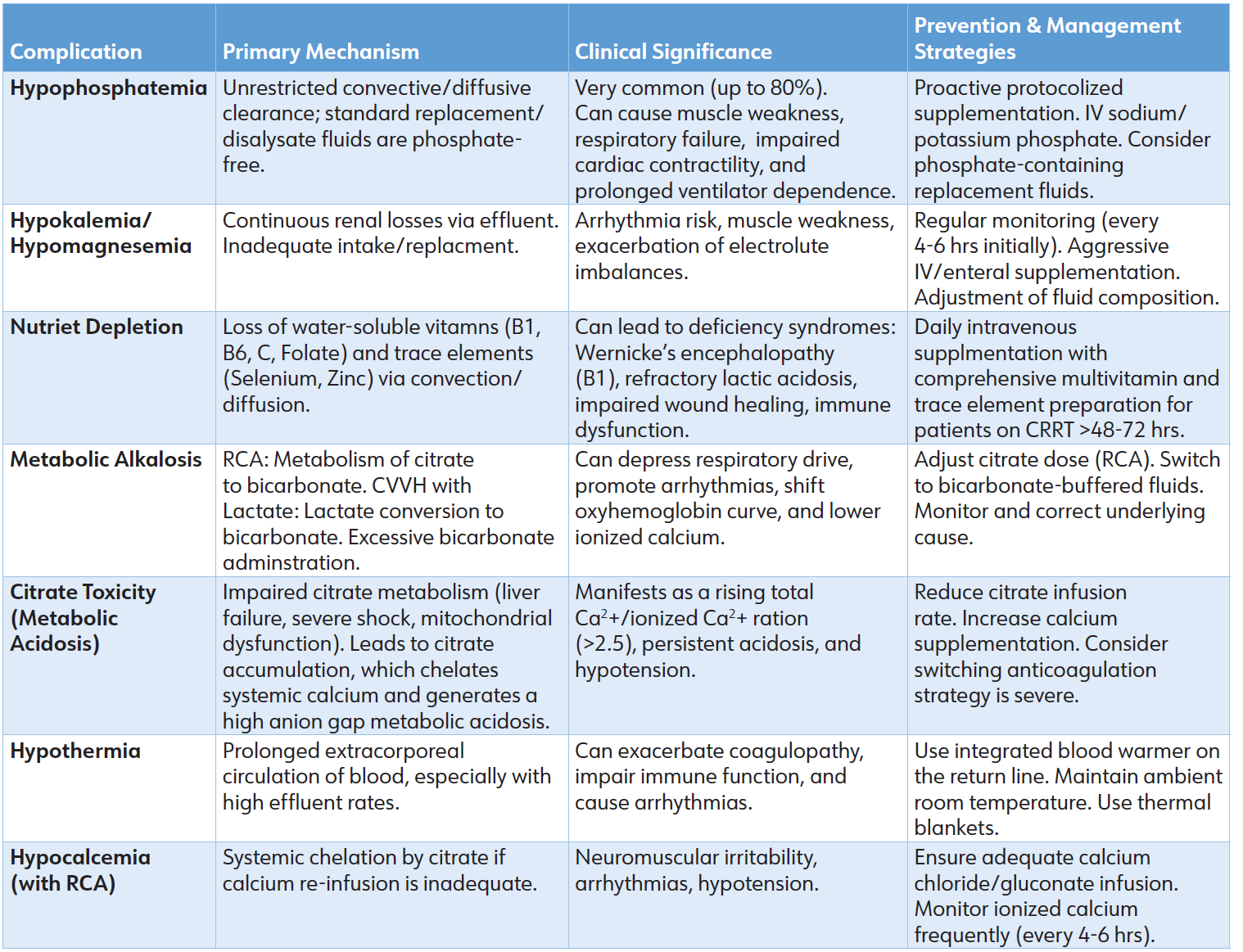
Abbreviations: CRRT: Continuous Renal Replacement Therapy; RCA: Regional Citrate Anticoagulation; CVVH: Continuous Venovenous Hemofiltration; IV: Intravenous
Vascular Access and Hemodynamic Complications
• Catheter-Related Bloodstream Infection (CRBSI): A significant contributor to morbidity. The risk associated with femoral catheters increases significantly with extended usage. Careful insertion technique, application of chlorhexidine-impregnated dressings, and daily assessment of catheter necessity are essential preventive measures. [44]
• Vascular Injury: Encompasses thrombosis, stenosis, or inadvertent arterial puncture occurring during insertion.
• Hemodynamic Instability: While CRRT offers greater hemodynamic stability compared to IHD, the risk of hypotension remains at the onset, especially when an excessive net negative fluid balance is implemented or in patients exhibiting significantly reduced vascular tone.
The dosing of medication during CRRT is highly intricate. The removal of drugs is influenced by various factors, including the properties of the drug (such as molecular weight, protein binding, and volume of distribution), patient-specific characteristics, and parameters related to CRRT. [45]
Principles of Drug Removal
The continuous removal of medications is a critical consideration for treatment. Water-soluble antibiotics with low protein binding, such as beta-lactams, vancomycin, and fluconazole, demonstrate significant clearance. [45] Standard intermittent dosing regimens often result in subtherapeutic concentrations, heightening the likelihood of treatment failure and the emergence of antimicrobial resistance. Therapeutic drug monitoring (TDM) plays a crucial role in informing dosing strategies, and it is often necessary to implement extended or continuous infusions to achieve and sustain therapeutic targets throughout the dosing interval. [45]
Dosing Strategies for Common Drug Classes
• Drugs with High Clearance: Drugs characterized by high clearance include water-soluble, low-proteinbinding agents with a small volume of distribution, which are removed most effectively. Examples include beta-lactams, vancomycin, fluconazole, and levetiracetam.
• Dosing Strategies: 1. Loading Dose: Should generally be unchanged. 2. Maintenance Dose: Typically necessitates a substantial increase in dosage or frequency. Timedependent antibiotics, such as beta-lactams, often require extended or continuous infusions.
• The Imperative of TDM is crucial for optimizing treatment and mitigating toxicity associated with medications that possess a narrow therapeutic index, such as vancomycin, aminoglycosides, and certain antiepileptics.
• Resources and Consultation: Dosing must be informed by established pharmacokinetic studies, institutional protocols, and collaboration with a clinical pharmacist.
Analgesics, Sedatives, and Vasoactive Agents
• Analgesics and sedatives, such as fentanyl, midazolam, and propofol, exhibit high protein binding or lipid solubility, resulting in minimal clearance by CRRT. Dosing is determined by clinical effect.
• Vasoactive Medications: Vasoactive medications, such as norepinephrine and vasopressin, exhibit minimal removal, with dosing adjusted to meet hemodynamic objectives.
CRRT significantly disrupts the homeostasis of nutrients, electrolytes, and pharmaceuticals. The ongoing removal of plasma water requires a proactive and anticipatory strategy for metabolic support and pharmacotherapy. [46]
Nutritional Support in CRRT
Patients with critical illness undergoing CRRT exhibit hypercatabolic and hypermetabolic states. CRRT exacerbates this by contributing continuous convective and diffusive losses.
• Protein and Amino Acid Requirements: International guidelines currently advocate for a high protein intake of 1.5 to 2.5 g/kg/day, utilizing adjusted or ideal body weight, to offset losses and promote anabolism. [42]
• Energy Requirements: Must be determined using indirect calorimetry when feasible. In the absence of this, predictive equations ranging from 25 to 30 kcal/ kg/day are utilized. [42]
• Trace Elements and Water-Soluble Vitamins: CRRT effectively removes water-soluble vitamins, particularly thiamine (B1), pyridoxine (B6), and ascorbic acid (C), as well as trace elements such as selenium and zinc. Daily intravenous supplementation with a comprehensive multivitamin and trace element preparation is standard care for patients anticipated to undergo CRRT for more than 48 to 72 hours to avert deficiency syndromes.
• Electrolyte Management: Proactive management of hypophosphatemia, hypokalemia, and hypomagnesemia is essential, often necessitating aggressive intravenous or enteral supplementation.
Despite extensive clinical application over several decades, CRRT continues to be characterized by considerable controversies and swift technological advancements. [47] Persistent Clinical Controversies
• The Optimal Timing Revisited – A Precision Approach: The current frontier involves personalizing decision-making through biomarker-defined subphenotypes of AKI to pinpoint patients who are most likely to benefit from earlier initiation of RRT, extending beyond the use of creatinine and urine output alone. [48]
• Anticoagulation in Extreme Physiology: The safety and efficacy of RCA in patients with severe hepatic failure or profound lactic acidosis remain debated, weighing the risk of citrate toxicity against the bleeding risk associated with alternative treatments.
• CRRT for Non-Renal Indications: The indications of high-volume hemofiltration or adsorptive therapies for inflammation modulation in sepsis are still under investigation, showing no consistent mortality benefit for unselected populations. [23,24] Future applications rely on improved patient stratification and enhanced targeted technologies.
Technological and Operational Innovations
• Advanced Membranes and Adsorptive Therapies: The advancement of sophisticated membranes and adsorptive technologies marks a crucial boundary in extracorporeal therapy, striving to progress from non-selective solute clearance to more precise blood purification methods. Commercially available hybrid membranes integrate traditional high-flux filtration with embedded adsorption materials, such as polymyxin B for endotoxin removal or specific resins targeting cytokines. Additionally, specialized adsorber cartridges employed in conjunction with a CRRT circuit have the capability to selectively eliminate inflammatory mediators or endotoxin. These technologies present a theoretical potential for influencing the dysregulated host response in scenarios such as septic shock or severe pancreatitis. Nonetheless, although encouraging studies and case series indicate enhancements in hemodynamic parameters and decreases in catecholamine needs, robust evidence from extensive RCTs showing a definitive mortality advantage in unselected populations is still lacking. [49,50] Present applications frequently rely on local protocols and the expertise of physicians, with their use generally regarded in cases of refractory shock despite conventional treatment.
The Role of Artificial Intelligence and Machine Learning
The incorporation of Artificial Intelligence (AI) and Machine Learning (ML) in CRRT management signifies a transition from static, protocol-based care to a more dynamic, predictive, and individualized approach. These technologies are under investigation in various critical domains:
• Predictive Analytics for Circuit Longevity: Predictive analytics utilizing machine learning models are under investigation to assess real-time data from the CRRT machine to predict imminent filter clotting several hours in advance, enabling preemptive interventions to extend circuit longevity and minimize downtime. [51]
• Precision Dosing and Dynamic Prescription: AI algorithms show promise in utilizing continuous data streams to suggest patient-specific adjustments to effluent doses, surpassing the fixed 25 mL/ kg/h standard. Machine learning can enhance the optimization of complex drug dosing in real-time by integrating CRRT parameters and TDM outcomes. [51]
• Clinical Decision Support for Initiation and Management: Advanced algorithms in early development are using extensive electronic health record datasets to detect intricate, non-linear patterns that indicate the necessity for RRT. These tools offer risk scores for AKI progression and recommend optimal initiation timing. [51]
• Automated Fluid Management and Alarm Integration: Theoretically, integrating intake and output data from various sources enables AI systems to deliver real-time net balance calculations and predictive alerts for fluid overload or depletion, thereby minimizing nursing cognitive load. The theoretical potential of AI and ML resides in their capacity to process multidimensional data and reveal patterns that are not detectable through conventional analysis. Key challenges for implementation involve ensuring robust, bias-free training datasets, achieving clinical interpretability (i.e., “explainable AI”), and integrating these tools into clinical workflows without contributing to alert fatigue.
• Wearable and Bioartificial Devices: Investigations into wearable artificial kidneys, focusing on miniaturization and sorbent regeneration, as well as bioartificial kidneys that incorporate living renal cells, illustrate a long-term goal for mobile and functional organ replacement. [52]
Global Equity and Sustainable Practice
A significant issue is the substantial global disparity in access to CRRT. Future directions should encompass the creation of cost-effective, streamlined platforms, allocation of resources for training, and investigation into viable intermediate therapies appropriate for resource-constrained environments.
This review consolidates the current evidence and practices surrounding CRRT. The foundational concept of ongoing, physiologically considerate adjustment has been solidly established. The accumulation of evidence supporting a standard effluent dose of 20 to 25 mL/kg/h, along with the significant preference for regional citrate anticoagulation, signifies important evidence-based progress. [13,30,31]
This review highlights a central paradox in contemporary CRRT practice: despite its technological sophistication, the overarching principle evidenced by major trials is one of measured restraint. The perspective has evolved from considering CRRT as a disease-modifying intervention to be started promptly and aggressively, to understanding it as a nuanced supportive mechanism to be utilized thoughtfully. This is particularly clear in the agreement on timing—where a “watchful waiting” strategy guided by explicit clinical criteria has replaced early empirical initiation—and in dosing, where a standard intensity is non-inferior to higher doses. The existing standard of care effectively weighs the significant capabilities of the technology against the crucial objective of preventing iatrogenic complications, all while delivering vital organ support.
The most notable advancement is the clarification of the “early vs. late” initiation debate, which now supports a “watchful waiting” approach informed by established absolute indications. [13,27,53] This transition highlights CRRT as an adjunctive measure for addressing the complications associated with AKI, rather than a treatment that alters the disease course. At the same time, management has evolved to require the proactive replacement of electrolytes, trace elements, and vitamins to avert iatrogenic harm. [17,41,42]
Ongoing controversies highlight the persistent gap between physiological understanding and clinical evidence. The lack of consistent improvement in mortality rates from blood purification strategies in sepsis indicates that the existing CRRT technology might be an inadequate tool for influencing a multifaceted immunologic response. [10,11]
Anticipating the future, the path is directed towards enhanced personalization and seamless integration. The future involves leveraging multimodal data to inform a therapy that is dynamically tailored in terms of dose, timing, and mechanism to align with the individual patient’s pathophysiology. [16,17,50]
Limitations
This review presents certain limitations that include
i. Methodological Constraints: As a narrative review designed for comprehensive clinical synthesis, a formal systematic review protocol was not employed. However, a targeted and structured literature search was conducted to ensure the inclusion of pivotal evidence. The selection and interpretation of evidence may be influenced by author bias.
ii. Evolving Evidence Base: Conclusions may be updated by later studies, especially in rapidly advancing fields such as immunomodulation and AI.
iii. Generalizability of Evidence: High-quality evidence is derived from well-equipped ICUs. The applicability in settings with limited resources is significantly restricted.
iv. Concentration on CRRT: An in-depth comparative examination with IHD and sustained low-efficiency dialysis (SLED) exceeds the parameters of this study.
v. Technological Heterogeneity: Distinct operational specifics may differ among various manufacturers and generations of machines.
vi. Scope and Depth: This broad narrative review seeks clinical comprehensiveness; however, an in-depth analysis of specific specialized applications—such as CRRT in pediatric populations or during concurrent extracorporeal membrane oxygenation (ECMO)— was beyond the scope of this article. Clinicians are encouraged to consult specialized guidelines for detailed management protocols in these specific domains.
CRRT has developed into a crucial and intricate method of life support in today’s ICUs. Its principal advantage lies in providing a physiologically gentle and continuous regulation of solute and fluid balance. Achieving mastery requires an in-depth grasp of its complex challenges: preventing electrolyte and nutrient depletion, skillfully navigating altered drug pharmacokinetics, and managing complications with vigilance. The discipline has evolved to acknowledge CRRT as an advanced supportive instrument. The future of CRRT is characterized by a growing emphasis on personalization and the integration of advanced technology, featuring precise timing, targeted therapies, and automated optimization. A concerted global effort is required to develop sustainable and accessible renal support solutions to tackle significant healthcare disparities. For the clinician, a profound understanding of the principles of CRRT grounded in physiology is crucial for implementing current best practices and ethically incorporating future innovations, ultimately aimed at enhancing survival and recovery for patients experiencing severe AKI and multiorgan failure.
AUTHORS’ CONTRIBUTION
All authors have significantly contributed to the work, whether by conducting literature searches, drafting, revising, or critically reviewing the article. They have given their final approval of the version to be published, have agreed with the journal to which the article has been submitted, and agree to be accountable for all aspects of the work.
SOURCE OF FUNDING
None.
CONFLICT OF INTEREST
None.
References
1. Ronco C, Bellomo R, Kellum JA. Acute kidney injury. Lancet. 2019;394(10212):1949-1964.
2. Mehta RL, Cerdá J, Burdmann EA, Tonelli M, García-García G, Jha V, et al. International Society of Nephrology’s 0by25 initiative for acute kidney injury (zero preventable deaths by 2025): A human rights case for nephrology. Lancet. 2015;385(9987):2616-2643.
3. Hoste EAJ, Bagshaw SM, Bellomo R, Cely CM, Colman R, Cruz DN, et al. Epidemiology of acute kidney injury in critically ill patients: The multinational AKI-EPI study. Intensive Care Med. 2015;41(8):1411-1423.
4. Hoste EAJ, Kellum JA, Selby NM, Zarbock A, Palevsky PM, Bagshaw SM, et al. Global epidemiology and outcomes of acute kidney injury. Nat Rev Nephrol. 2018;14(10): 607-625.
5. Uchino S, Bellomo R, Goldsmith D, Bates S, Ronco C. An assessment of the RIFLE criteria for acute renal failure in hospitalized patients. Crit Care Med. 2006;34(7): 1913-1917.
6. Post EH, Kellum JA, Bellomo R, Vincent JL. Renal perfusion in sepsis: From macro- to microcirculation. Kidney Int. 2017;91(1):45-60.
7. Perazella MA, Rosner MH. Drug-induced acute kidney injury. Clin J Am Soc Nephrol. 2022;17(8):1220-1233.
8. Palevsky PM, Liu KD, Brophy PD, Chawla LS, Parikh CR, Thakar CV, et al. KDOQI US commentary on the 2012 KDIGO clinical practice guideline for acute kidney injury. Am J Kidney Dis. 2013;61(5):649-672.
9. Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. 2024;105(4S):S117-S314.
10. Hoste EA, McCullough PA, Kashani K, Chawla LS, Joannidis M, Shaw AD, et al; Sapphire Investigators. Derivation and validation of cutoffs for clinical use of cell cycle arrest biomarkers. Nephrol Dial Transplant. 2014; 29(11):2054-2061.
11. Ostermann M, Zarbock A, Goldstein S, Kashani K, Macedo E, Murugan R, et al. Recommendations on Acute Kidney Injury Biomarkers from the Acute Disease Quality Initiative Consensus Conference: A consensus statement. JAMA Netw Open. 2020;3(10):e2019209.
12. Chawla LS, Eggers PW, Star RA, Kimmel PL. Acute kidney injury and chronic kidney disease as interconnected syndromes. N Engl J Med. 2014;371(1):58-66.
13. Bottari G, Ranieri VM, Ince C, Pesenti A, Aucella F, Scandroglio AM, et al. Use of extracorporeal blood purification therapies in sepsis: The current paradigm, available evidence, and future perspectives. Crit Care. 2024;28(1):432.
14. Tandukar S, Palevsky PM. Continuous renal replacement therapy: Who, when, why, and how. Chest. 2019;155(3): 626-638.
15. Lang T, Zawada AM, Theis L, Braun J, Ottillinger B, Kopperschmidt P, et al. Hemodiafiltration: Technical and medical insights. Bioengineering (Basel). 2023;10(2):145.
16. Villa G, Neri M, Bellomo R, Cerda J, De Gaudio AR, De Rosa S, et al. Nomenclature for renal replacement therapy and blood purification techniques in critically ill patients: Practical applications. Crit Care. 2016;20(1):283.
17. Murugan R, Hoste E, Mehta RL, Samoni S, Ding X, Rosner MH, et al; Acute Disease Quality Initiative (ADQI) Consensus Group. Precision fluid management in continuous renal replacement therapy. Blood Purif. 2016; 42(3):266-278.
18. Gaudry S, Palevsky PM, Dreyfuss D. Extracorporeal kidney-replacement therapy for acute kidney injury. N Engl J Med. 2022;386(10):964-975.
19. Bart BA, Goldsmith SR, Lee KL, Givertz MM, O’Connor CM, Bull DA, et al; Heart Failure Clinical Research Network. Ultrafiltration in decompensated heart failure with cardiorenal syndrome. N Engl J Med. 2012;367(24): 2296-2304.
20. Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. KDIGO clinical practice guideline for acute kidney injury. Kidney Int Suppl. 2012; 2(1):1-138.
21. Chen H, Yu RG, Yin NN, Zhou JX. Combination of extracorporeal membrane oxygenation and continuous renal replacement therapy in critically ill patients: A systematic review. Crit Care. 2014;18(6):675.
22. Ostermann M, Joannidis M, Pani A, Floris M, De Rosa S, Villa G, et al. Patient selection and timing of continuous renal replacement therapy. Blood Purif. 2016;42(3):224-237.
23. Pickkers P, Vassiliou T, Liguts V, Prato F, Tissieres P, Kloesel S, et al. Sepsis management with a blood purification membrane: European experience. Blood Purif. 2019;47 Suppl 3:1-9.
24. Gaudry S, Hajage D, Benichou N, Chaïbi K, Barbar S, Zarbock A, et al. Delayed versus early initiation of renal replacement therapy for severe acute kidney injury: A systematic review and individual patient data metaanalysis of randomised clinical trials. Lancet. 2020; 395(10235):1506-1515.
25. Bagshaw SM, Wald R, Adhikari NKJ, Bellomo R, da Costa BR, Dreyfuss D, et al. Timing of initiation of renalreplacement therapy in acute kidney injury. N Engl J Med. 2020;383(3):240-251. Erratum in: N Engl J Med. 2020;383(5):502.
26. Zarbock A, Kellum JA, Schmidt C, Van Aken H, Wempe C, Pavenstädt H, et al. Effect of early vs delayed initiation of renal replacement therapy on mortality in critically ill patients with acute kidney injury: The ELAIN randomized clinical trial. JAMA. 2016;315(20):2190-2199.
27. Bourgeois S, Joannes-Boyau O, Honore PM, De Waele E, Vanbelleghem H, Collin V, et al. Technical aspects of continuous renal replacement therapy. In: Critical Care Nephrology. 3rd ed. Elsevier; 2019. p.888-894.e1.
28. Palevsky PM, Zhang JH, O’Connor TZ, Chertow GM, Crowley ST, Choudhury D, et al. Intensity of renal support in critically ill patients with acute kidney injury. N Engl J Med. 2008;359(1):7-20.
29. Bellomo R, Cass A, Cole L, Finfer S, Gallagher M, Lo S, et al. Intensity of continuous renal-replacement therapy in critically ill patients. N Engl J Med. 2009;361(17): 1627-1638.
30. Vesconi S, Cruz DN, Fumagalli R, Kindgen-Milles D, Monti G, Marinho A, et al; DOse REsponse Multicentre International collaborative Initiative (DO-RE-MI Study Group). Delivered dose of renal replacement therapy and mortality in critically ill patients with acute kidney injury. Crit Care. 2009;13(2):R57.
31. Liu C, Mao Z, Kang H, Hu J, Zhou F. Regional citrate versus heparin anticoagulation for continuous renal replacement therapy in critically ill patients: A metaanalysis with trial sequential analysis of randomized controlled trials. Crit Care. 2016;20(1):144.
32. Li R, Gao X, Zhou T, Li Y, Wang J, Zhang P. Regional citrate versus heparin anticoagulation for continuous renal replacement therapy in critically ill patients: A metaanalysis of randomized controlled trials. Ther Apher Dial. 2022;26(6):1086-1097.
33. Gattas DJ, Rajbhandari D, Bradford C, Buhr H, Lo S, Bellomo R, et al. A randomized controlled trial of regional citrate versus regional heparin anticoagulation for continuous renal replacement therapy in critically ill adults. Crit Care Med. 2015;43(8):1622-1629.
34. Bai M, Zhou M, He L, Ma F, Li Y, Yu Y, et al. Citrate versus heparin anticoagulation for continuous renal replacement therapy: An updated meta-analysis of RCTs. Intensive Care Med. 2015;41(12):2098-2110.
35. Legrand M, Tolwani A. Anticoagulation strategies in continuous renal replacement therapy. Semin Dial. 2021; 34(6):416-422.
36. Hu C, Shui P, Zhang B, Xu X, Wang Z, Wang B, et al. How to safeguard the continuous renal replacement therapy circuit: A narrative review. Front Med (Lausanne). 2024;11:1442065.
37. Schmidt M, Pellegrino V, Combes A, Scheinkestel C, Cooper DJ, Hodgson C. Mechanical ventilation during extracorporeal membrane oxygenation. Crit Care. 2014; 18(1):203.
38. Joannidis M, Druml W, Forni LG, Groeneveld ABJ, Honore PM, Hoste E, et al. Prevention of acute kidney injury and protection of renal function in the intensive care unit: Update 2017: Expert opinion of the Working Group on Prevention, AKI section, European Society of Intensive Care Medicine. Intensive Care Med. 2017;43(6):730-749.
39. Samoni S, Husain-Syed F, Villa G, Ronco C. Continuous renal replacement therapy in the critically ill patient: From garage technology to artificial intelligence. J Clin Med. 2021;11(1):172.
40. Douvris A, Zeid K, Hiremath S, Bagshaw SM, Wald R, Beaubien-Souligny W, et al. Mechanisms for hemodynamic instability related to renal replacement therapy: A narrative review. Intensive Care Med. 2019; 45(10):1333-1346.
41. Thompson Bastin ML, Stromberg AJ, Nerusu SN, Liu LJ, Mayer KP, Liu KD, et al. Association of phosphatecontaining versus phosphate-free solutions on ventilator days in patients requiring continuous kidney replacement therapy. Clin J Am Soc Nephrol. 2022;17(5):634-642.
42. Berger MM, Shenkin A, Schweinlin A, Amrein K, Augsburger M, Biesalski HK, et al. ESPEN micronutrient guideline. Clin Nutr. 2022;41(6):1357-1424.
43. Jacobs R, Verbrugghe W, Dams K, Roelant E, Couttenye MM, Devroey D, et al. Regional citrate anticoagulation in continuous renal replacement therapy: Is metabolic fear the enemy of logic? A systematic review and metaanalysis of randomised controlled trials. Life (Basel). 2023;13(5):1198.
44. Buetti N, Marschall J, Drees M, Fakih MG, Hadaway L, Maragakis LL, et al. Strategies to prevent central line-associated bloodstream infections in acute-care hospitals: 2022 Update. Infect Control Hosp Epidemiol. 2022;43(5):553-569.
45. Li L, Li X, Xia Y, Chu Y, Zhong H, Li J, et al. Recommendation of antimicrobial dosing optimization during continuous renal replacement therapy. Front Pharmacol. 2020; 11:786.
46. O’Dell Duplechin M, Folds GT, Duplechin DP, Ahmadzadeh S, Myers SH, Shekoohi S, et al. Prevention and management of perioperative acute kidney injury: A narrative review. Diseases. 2025;13(9):295.
47. Gist KM, Fuhrman DY, Deep A, Haga T, Demirkol D, Bell MJ, et al. Continuous renal replacement therapy: Current state and future directions for worldwide practice. Pediatr Crit Care Med. 2024;25(6):554-560.
48. Bhatraju PK, Zelnick LR, Himmelfarb J. Precision medicine in acute kidney injury: Biomarker-defined subphenotypes and their implications for renal replacement therapy timing. Nat Rev Nephrol. 2024;20(2):85-99.
49. Snow TAC, Littlewood S, Corredor C, Singer M, Arulkumaran N. Effect of extracorporeal blood purification on mortality in sepsis: A meta-analysis and trial sequential analysis. Blood Purif. 2021;50(4-5): 462-472.
50. Putzu A, Schorer R, Lopez-Delgado JC, Cassina T, Landoni G. Blood purification and mortality in sepsis and septic shock: A systematic review and meta-analysis of randomized trials. Anesthesiology. 2019;131(3): 580-593.
51. Qiu S, Mu S, Tao Y, Zhang N, Bai J, Cao N. Machine learning model predicts clotting risk during CRRT in ESKD patients: A SHAP-interpretable approach. Ren Fail. 2025;47(1):2562448.
52. Dolson GM. The wearable artificial kidney. Methodist Debakey Cardiovasc J. 2020;16(4):324-325.
53. Joannidis M, Forni LG, Klein SJ, Honore PM, Kashani K, Ostermann M, et al. Lung-kidney interactions in critically ill patients: Consensus report of the Acute Disease Quality Initiative (ADQI) 21 Workgroup. Intensive Care Med. 2020;46(5):654-672.
